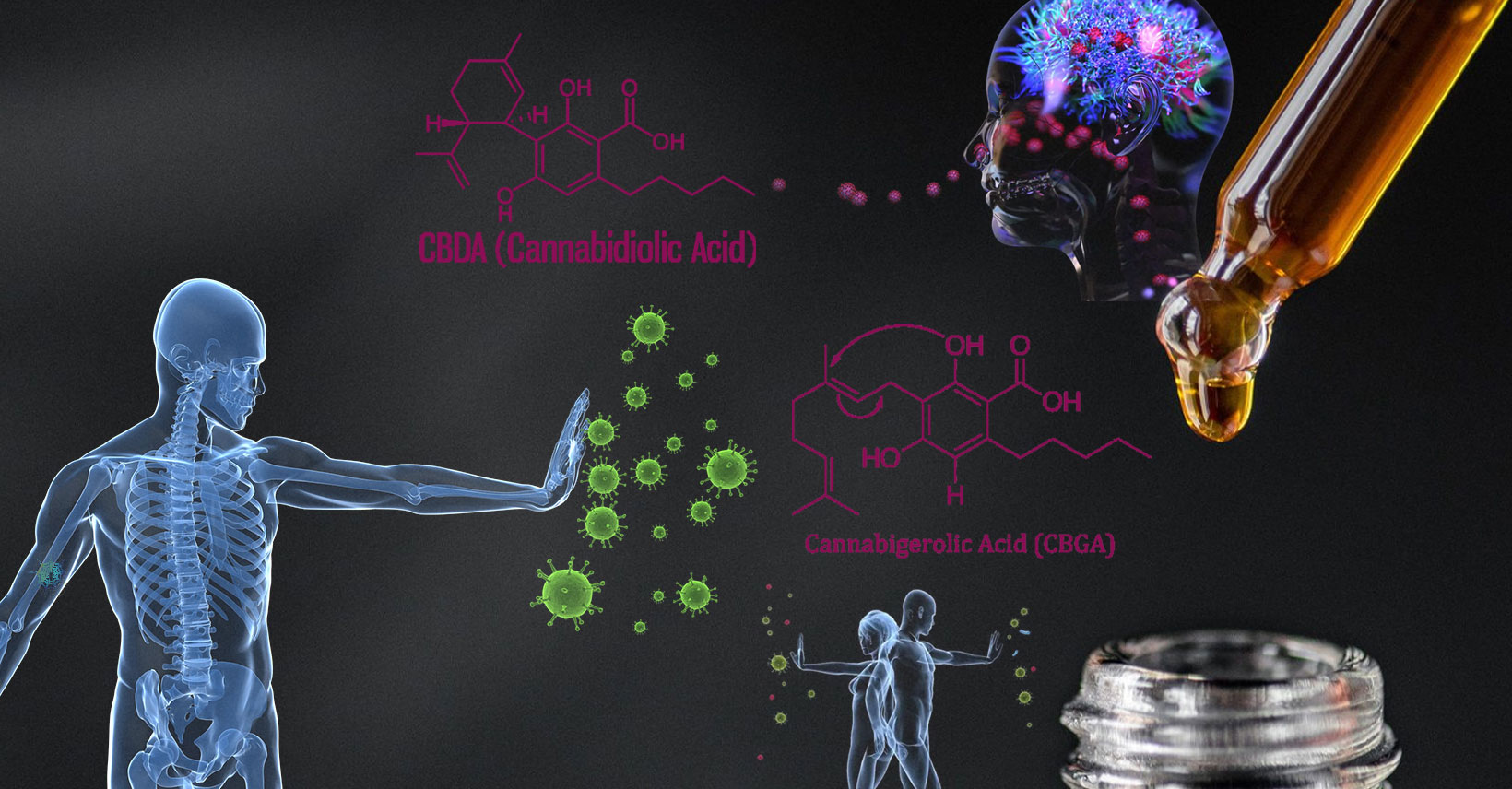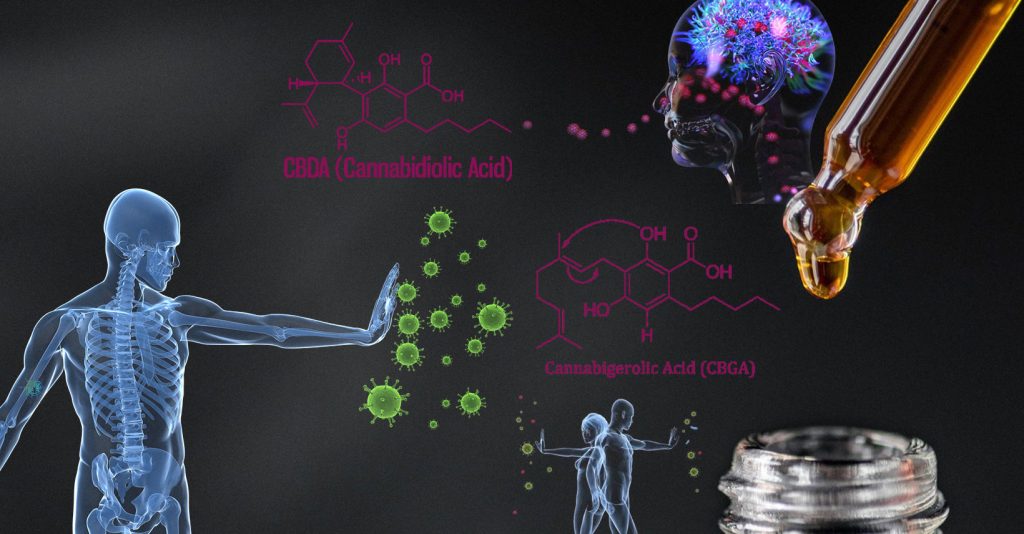
CBGA & CBDA may Block the COVID-19 Virus, yet even with this OSU report, the FDA will consider that product an unapproved drug.
Recently researchers at Oregon State University found that cannabigerolic acid (CBGA) & cannabidiolic acid (CBDA) “bind to the SARS-CoV-2 spike protein, blocking a critical step in the process the virus uses to infect people.” I am so grateful for these students. Their hard work has given way to some research that may take us over the precipice for the federal legalization of plant medicine.
Richard van Breemen, leader of this study, has this to say. “Any part of the infection and replication cycle is a potential target for antiviral intervention, and the connection of the spike proteins receptor-binding domain to the human cell surface receptor ACE2 is a critical step in that cycle.” But what does this mean, Richard?
Entitled “Cannabinoids Block Cellular Entry of SARS-CoV-2 and the Emerging variants,” researchers published their findings in a research paper on January 11, 2022.
“That means cell entry inhibitors, like the acids from hemp, could be used to prevent SARS-CoV-2 infection and also to shorten infections by preventing virus particles from infecting human cells. They bind to the spike proteins, so those proteins can not bind to the ACE2 enzyme, which is abundant on the outer membrane of endothelial cells in the lungs and other organs.”
Final thoughts:
Not everyone can keep up with a conversation once you start tossing around terms like spike proteins, ACE2 enzyme, and SARS-CoV-2. I tend to tune it all out as the topic is a little too real for jCruceWeb, but this topic holds a special place in my canna heart. Is this the final piece of the puzzle that will loosen the grips of federal prohibition and allow for the healing to begin? It leaves me wondering, does it matter that the FDA approves this essential cannabinoid?